 
Neutral oxides have neither acidic not basic properties. The amphoteric oxides show both acidic and basic properties. 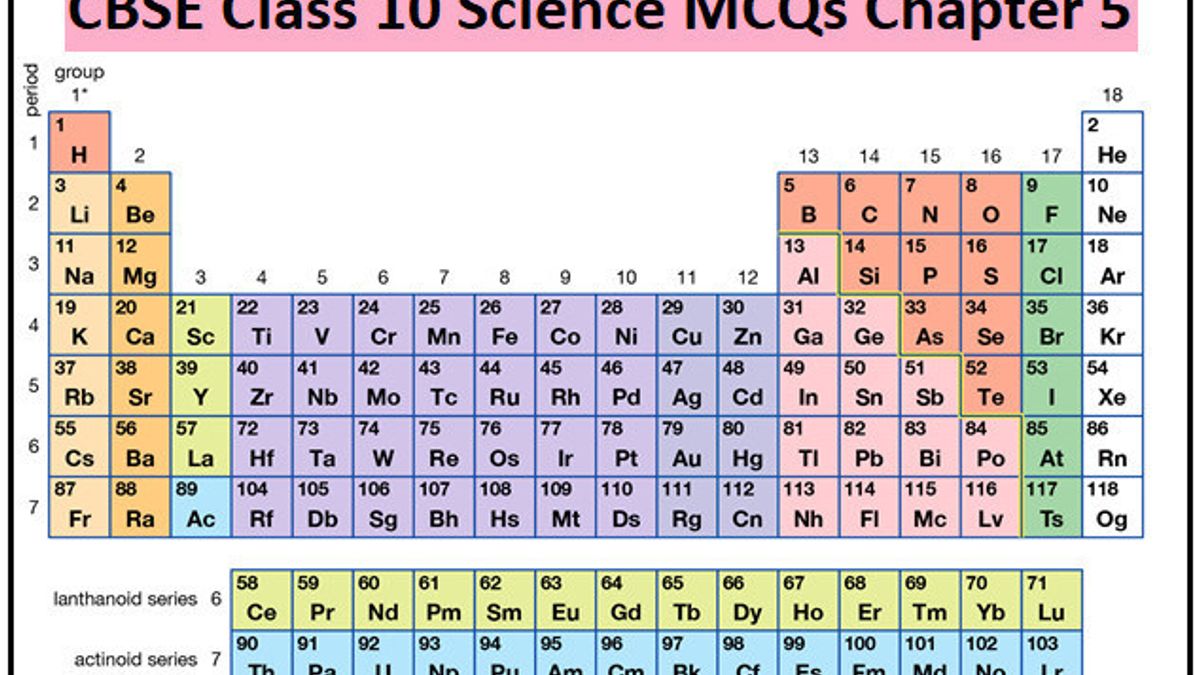
Oxides of the elements in the centre are either amphoteric or neutral. The elements at the extreme left of the periodic table readily combine with oxygen to form oxides which are most basic.Įlements on extreme right from oxides which are most acidic. Elements are organized on the table according to their atomic number. Metallic character of an element which is highest at the extreme left decreases while the non metallic character increases on moving across the period from left to right. Organizes the elements by chemical properties. The chemical reactivity of alkali metals on the extreme left is due to their ability to lose an electron to form corresponding cation.Ĭhemical reactivity of halogens on the extreme right is due to their ability to gain an extra electron leading to the formation of the anion.Īlkali metals are good reducing agents while halogens are good oxidizing agents. 6-Periodic Table periodictable.pdf 17 kb pdf 38 kb doc notes-periodictrendcharts.doc 358. There is high chemical reactivity at the two extremes and lowest in the centre. Starting from lithium (Li), the eighth element is sodium(Na) and its properties are similar. Ionization enthalpy of the extreme left element in a period is the lowest while electron gain enthalpy of the element at the extreme right is the most negative. The arrangement of elements given by Newlands is given in Table 6.2. It contains substances ranging from Radon to Cesium. As a result ionization enthalpy generally increases and electron gain enthalpy become more negative across a period. The sixth period in the periodic table is regarded as the longest period. The atomic and ionic radii decreases as we move across the period from left to right. (ii) Position of isotope: Isotopes should be placed separately according to periodic law.But actually one place was given to all isotopes of an element. The chemical and physical properties of the elements depend mainly upon their electronic configuration. Read also: Chemical Bonding and Molecular Structure Class 11 Notes Chemistry Chapter 4 Defects in Mendeleev’s Table (i) Position of hydrogen: Position of hydrogen was not justified. When we move down the group, the number of valence electrons remain the same, therefore all the elements in a group exhibit the same valence.ĥ) the maximum covalency of first member of each group is 4 whereas other members of the groups can have a maximum covalency of 6.Ħ) because of smaller size and high electronegativity ,first member of each group show greater ability to form pπ-pπ multiple bonds either with itself or with other members of the second period. The oxidation state of an element in a given compound may be defined as the charge acquired by its atom on the basis of electronegativity of the other atoms in the molecule. Transition and inner transition elements, exhibit variable valency due to the involvement of not only the valence electrons but the d or f electrons as well.Īs we move across a period from left to right, the number of valence electron increases from 1 to 8. THE PERIODIC TABLE Addresses part of Massachusetts Curriculum Frameworks, content standard 3. The valence of an atom equal to either the number of valence electrons are equal to 8 minus the number of valence electron. The orbitals present in the valence shell are called valence orbitals. The electrons present in the outermost shell of an atom are called valence electrons and the number of these electrons determine the valence or the valency of the atom.  
Conduct heat and electricity Metalloids B, Si, As, Te, At, Ge, Sb Metalloids look like stairs share properties with metals and nonmetals Halogens contain solids gases and liquids.Periodicity of valence or Oxidation State Ductile able to be formed into a wire Transition Metals Harder, more brittle, higher melting point than groups 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
